Beef Fat Extract Beef Fat Extract Ole

Rendering of Beef Tallow for Biodiesel Product: Microwave versus Boiling Water and Acetone Fat Extraction
i
Instituto Superior Técnico, Departamento de Engenharia Química, Universidade de Lisboa, Av. Rovisco Pais, 1, 1049-001 Lisboa, Portugal
2
CERENA, Instituto Superior Técnico, Universidade de Lisboa, Av. Rovisco Pais, i, 1049-001 Lisboa, Portugal
*
Author to whom correspondence should be addressed.
Bookish Editor: Chiing-Chang Chen
Received: six March 2022 / Revised: 23 March 2022 / Accustomed: 27 March 2022 / Published: 29 March 2022
Abstract
Biodiesel can substitute for conventional diesel fuel and contribute to the decarbonization of the transportation sector. To improve biodiesel sustainability and decrease production costs, low-grade fats such equally non-edible beast fats must be used. Fauna fats are mixed with tissues which must exist removed before alcoholysis to avert biodiesel contamination with nitrogen and phosphorus-containing compounds. Biodiesel was produced by the methanolysis of beefiness tallow and beef tallow/soybean oil mixtures over calcium heterogeneous catalysts obtained by the calcination of scallop shells. The tallow from fatty bovine tissues was extracted using boiling water, dry microwave handling, and acetone extraction. The thermal stability and the wet content of the extracted fats were evaluated by thermogravimetry. The thermograms of fats revealed that microwave treatment, which was faster (iii min instead of 40 min for boiling water and 240 min for acetone extraction) and had the lowest free energy consumption, led to a dry fatty with a thermal stability analogous to that of fats extracted with humid water and acetone. All the extracted fats behaved similarly in the methanolysis reaction over calcium catalyst, with biodiesel yield (61–62%) being thirty% lower than the coordinating obtained from soybean oil (88%). Co-processing the extracted tallow with soybean oil overcomes the drawback related to the depression-course fats.
1. Introduction
The mitigation of anthropogenic carbon can be partially attained past substituting fossil fuels with renewable ones. Biodiesel, a mixture of alkyl esters, has been pointed out as a feasible renewable fuel in the transportation sector [1,ii], which is the primary contributor to anthropogenic CO2. First-generation biodiesel, derived from vegetable oils, presents several sustainability issues [3]. The occupation of arable state with oil crops defended to fuel production is seen as a threat to nutrient production capacity, which triggers strong opposition to biodiesel due to fuel/food competition [4]. The price of vegetable oils is another negative factor as it makes the production of biodiesel expensive [5] and uncompetitive with the fossil fuel congener, except for very high prices of crude oil [6]. Bashir et al. [7] emphasized, in a recent review, that the choice of biodiesel fat feedstock is the well-nigh important gene in obtaining a sustainable and economically-competitive renewable transportation fuel. According to these researchers, the third- (algae and waste product oils) and fourth- (advanced solar oils) generation fat feedstocks are preferred in biodiesel production at a commercial scale.
Biodiesel product from animal fats has great potential because this feedstock does not compete with the food industry and helps with waste management [viii]. Animal fat is viewed equally a promising biodiesel raw material due to its low cost and easy availability [9]. Despite the advantages of biodiesel derived from creature fat, this raw material only makes upwardly seven% of the fat in biodiesel produced worldwide and but 4% of the biodiesel produced in EEC [10]
Beef tallow, pork lard, craven fat, and grease are some examples of animal fats that can be employed [eleven,12,13,14,fifteen,16,17]. Animal fatty feedstocks atomic number 82 to a biodiesel with higher cetane number than that of vegetable oils. The high content of saturated fatty acids is responsible for this improved characteristic of creature fat biodiesel [16]. Furthermore, the high cetane number of animal fat biodiesel allows for a reduction in NOX emissions [17]. Because that raw materials accept the greatest weight in the cost construction of biodiesel product (Figure one), the use of beast fats instead of vegetable oils contributes to the reduction in production costs. Animal fats are cheaper than vegetable oils and used cooking oils (Table 1). According to the literature, animate being fats (AF) contain high levels of free fat acids (FFA), which makes conventional biodiesel production impracticable due to soap formation. Complex processes to catechumen AF into biodiesel are reported in the literature. Ikura et al. [18] combined thermal great and acid-catalyzed esterification to produce biodiesel from animal fats. The process, afterward the thermal cracking, included a distillation process to separate the FFA for the esterification footstep. Simpler procedures, as schematized in Effigy 1, always include the pretreatment of creature fatty to subtract the fatty acidity [19].
Raw animal fat has protein-rich tissues which must be removed before the alcoholysis reaction. The fats are recovered from waste fat tissues by the rendering processes, mainly wet or dry rendering [20,21]. In wet rendering, the fat is recovered through heating in the presence of water. Boiling in h2o and/or steam at high temperatures can exist employed [21,22]. In dry rendering, the fatty tissues are cooked in their "own juices" with dry out heat [22].
Several processes are used in industry, all of them involving the application of heat, the extraction of wet, and the separation of fat.
The boiling water rendering presents some issues: high energy and time consumption, a fat with a high humidity, and increased free fatty acid content due to the long contact of fat with water [21]. Additionally, considering that the main purpose of the rendering is the separation of fat from other materials including residual moisture, the introduction of additional moisture is seen equally unproductive [21]. For these reasons, some processes were replaced by dry out ones.
In recent years, microwave technology (MW) has received a lot of attention from researchers who apply it in a wide range of processes such equally methane fermentation [23,24], sludge treatment [25], lipid extraction from microalgae [26], MW-assisted catalyzed reactions [27], and even non-catalyzed biodiesel production [28]. According to researchers, MW technology consumes less energy than traditional technologies and is thus more cost-constructive. Microwave (MW) technology can also be used for animal fat rendering. This process is quicker, energy efficient, and the fat-rendering yield obtained is higher [29]. Lin et al. [30] reported that, among all the rendering methods used to recover craven fatty from broilers, the MW rendering was the nigh promising ane, obtaining the highest rendering yields and quality. Sheu and Chen [31] reported similar results: MW produced the highest rendering yields and the lightest-colored broiler peel fats. Recently, Hewavitharana et al. [32] compared several methods of extracting fatty from alimentary products and concluded that microwave-assisted fatty extraction is an effective methodology when compared with conventional methods, including the Folch method (liquid extraction with chloroform plus methanol mixture). The authors underlined the short processing time for microwave-assisted fat extraction. The literature on the microwave-assisted extraction of animal fat is not abundant and the existing literature does not explain how microwaves human action in the solvent-free extraction of fat. Taking into account the mechanism described for the extraction of microalgae oils [33], it is assumed that microwaves heat the h2o molecules found in the adipose tissue, which promotes localized heating and increased pressure, causing the breakdown of adipocyte, and releasing triglycerides
.Conventional methods of fat extraction involve the apply of solvents. Such methods are often used in the extraction of oil from oilseeds [34,35]. In a previous report, the utilize of acetone as a co-solvent in the methanolysis reaction not merely improved components' miscibility just also avoided catalyst deactivation by the adsorption of oily species [36]. For these reasons, the rendering procedure using acetone as the solvent was also evaluated.
The post-obit sections give data regarding the manufacture of biodiesel using beef tallow in order to contribute to the body of knowledge in the area of sustainable biodiesel product from non-edible brute fat. To increase the fat yield that allows biodiesel production past methanolysis over heterogeneous bones catalysis (CaO goad), unlike methods of extracting beef tallow were investigated (humid water, extraction with acetone, and microwave-assisted). Depression energy consumption, depression moisture content, and low acidity of the extracted fat were the goals of the fat-rendering studied procedures. The methanolysis of alimentary grade soybean oil (SBO) was used every bit a standard. Calcium-based heterogeneous catalyst, obtained from waste product materials, was used due to its recognized catalytic performance in biodiesel production [37].
2. Materials and Methods
The experimental work followed the steps outlined in Figure 2, starting with the extraction of beef tallow, which was then used in the production of biodiesel, and ending with the quantification of biodiesel and the label of the co-produced glycerin.
The beefiness tallow collected from a local butcher was subject to several rendering processes (Figure ii) to dissever fatty from protein-rich tissues. In the microwave-assisted (MW) process, a plastic container with 100 g of beef tallow was placed in a microwave oven and heated at maximum power (1400 W) for ane, ii, and 3 min, separately. Later removing the solid residues, the liquid was cooled at room temperature.
For boiling water (BW) fat extraction, the fat tissues were boiled with water (enough to cover the fat material) in a pressure cooker for forty min (1.five atm; 112 °C). Subsequently removing the residues, the liquid was cooled at room temperature and the fat layer was removed with a drainer. The cooking time was optimized to excerpt the maximum fat.
Acetone fat extraction, at reflux temperature, was accomplished in a glass 500 mL round flask equipped with a reflux column heated past a nest-shaped heating drapery. The fat tissues were boiled with acetone (1.28 wfat/due westacetone ratio) for 4 h. Afterwards removing the solid material, the liquid was cooled at room temperature and the fat layer was removed with a drainer. All the extraction methods were replicated iii times to evaluate their reproducibility.
The thermal stability and moisture content of the extracted fats was assessed by thermogravimetry. The thermograms were acquired, under airflow (constructed, dry), for samples of lx–100 mg in alumina crucibles, using a Netzsch TG–DTA-DSC thermobalance and a heating rate of 20 °C/min. The thermal degradation contour of soybean oil was used equally a reference. The gratis fatty acid (FFA) content of the fatty feedstocks was assessed past alkalimetric titration according to the standard EN 14214:2003 [38,39].
Biodiesel was produced by the methanolysis of beef tallow and beef tallow/soybean oil (1:i in weight basis) over calcium oxide catalyst using standard conditions to examination calcium-based catalysts [40]. The methanolysis tests, at methanol reflux temperature, were carried out in a batch reactor, equally previously described [41]. Briefly, the catalytic tests were carried out in a 3-neck 500 mL drinking glass reactor using a nest-shaped heating mantle and a magnetic stirrer (orbital magnet). The slurry containing the methanol (methanol/fatty = 12 molar ratio) and the catalyst (5%, fatty weight basis) was heated to the reflux temperature of methanol, remaining at this temperature for 1 h to promote the activation of methanol molecules on the catalyst surface [42]. Acetone-assisted methanolysis tests (5methanol/Vacetone = two.8) were carried out as reported by Dias et al. [36], to avert the catalyst deactivation past fat acidity and moisture. At the end of this period, the fatty, previously heated to 67 °C, was added, starting the counting of the reaction fourth dimension. After the reaction flow (2.5 h), the goad was separated from the reaction mixture by filtration (paper filter course 1, 11 μm). The glycerin and the oily phases were separated by gravitational settling in a funnel. Both liquid phases were characterized by ATR-FTIR and the methyl esters were quantified equally described by Dias et al. [43]. Briefly, the FAME yield ciphering was fabricated using the infrared spectral features in the 1480–1410 cm−one range, attributing the band centered at 1436 cm−ane to FAME. A calibration curve was previously obtained for oil/FAME mixtures with different FAME contents. The infrared spectra were collected with a resolution of iv cm−1, using an FT-MIR Perkin Elmer Spectrum Two IR Spectrometer. A horizontal total attenuated reflection accompaniment (ATR), from PIKE Technologies, with a ZnSe crystal, was used. The catalytic test using MW tallow, which was randomly selected, was replicated 3 times to evaluate the reproducibility of the catalytic test.
The calcium catalyst (mainly CaO) was obtained past the calcination of calcium-rich seafood shells [44,45]. The scallop shells, which had been household waste, were washed using tap water and dried naturally for several days (approximately a calendar week) while in contact with the laboratory'south atmospheric air. The stale shells were coarsely crushed and calcined for 3 h in a conceal furnace at 800 °C (heating rate 5 °C/min) in a static air atmosphere. The calcined material, after cooling to room temperature, was finely ground using a mechanical agate mortar and sieved to remove the fraction above 175 μm. The as-prepared catalyst was characterized by XRD to identify crystalline phases and the degree of crystallinity, and by ATR-FTIR (using the above-described equipment and conditions) to characterize the surface Ca species equally previously described [46]. The diffractogram was recorded with a Rigaku Geigerflex diffractometer with Cu Kα radiation at 40 kV and 40 mA (two°/min). The XRD pattern, for Bragg angle 2θ in the range 5–70°, was compared with JCPDS standard files to identify the crystalline phases. The morphology of scallop shells, raw and calcined, was examined by scanning electron microscopy (SEM) using Joel JSM7001F FEG-SEM equipment. The powdered samples were spread over a carbon agglutinative tape and covered with a thin metallic film of Pd-Au to improve their electronic conductivity.
3. Results and Discussion
The SEM images (Effigy 3) of the scallop shells, before and subsequently calcination, show that the initial morphology, agglomerates of thin calcite lamellae, transforms into a more than disorganized morphology with cubic calcite crystallites, mixed with material, with irregular (in terms of both shape and size) crystallites belonging to the lime. Observed morphologies are similar to those reported in the literature for scallop shell-derived materials [47].
Characterization of the prepared catalyst by XRD revealed patterns belonging to lime contaminated with calcite (Effigy 4), which agrees with the crystallite morphologies observed by SEM. The XRD data show that the calcination temperature was too low to fully decompose the calcium carbonate, which is the master component of scallop shells [45]. No XRD lines of portlandite (JCPDS 01-072-0156) were detected.
The FTIR spectrum (Figure v) of the fresh goad, with the well-nigh intense ring centered at 3641 cm−1, shows that the catalyst surface is dominated by hydroxyl species [46], which are the agile sites of methanol activation, forming surface methoxy species [42]. The less intense bands, for wavenumbers below 1600 cm−1, belong to adsorbed CO2 and not-decomposed calcite (CaCOthree) [46].
All the extraction methodologies allowed for a clear and homogeneous fat, but with dissimilar fatty yields. The energy consumption and fat yield of each rendering process are summarized in Table 2. The microwave-assisted method was the nigh performant methodology in the extraction procedure since it presented the everyman power consumption per gram of extracted fat. The loftier potential of microwaves in the production of biodiesel, including the fat extraction steps, has been previously reported [48]. The authors also report that microwaves have lower energies than Brownian motion, thus are unable to break chemical bonds, which prevents the degradation of raw materials past unwanted reactions.
The boiling water process presented high free energy consumption and led to fat with a high moisture content, which is a disadvantage for biodiesel production by alcoholysis over calcium catalyst [46].
The fat extracted using humid water and acetone had a greater acidity than that extracted by MW. The boiling water can aid in the hydrolysis of triglycerides, resulting in a greater FFA level (Equation (1), [49]). For acetone-extracted fat, the loftier acidity is justified by the duration of the operation and the fact that acetone is not miscible with the glycerin produced during the hydrolysis of triglycerides, which prompts the hydrolysis.
The thermal stability of extracted beefiness tallow was assessed past thermogravimetry under airflow. The thermograms in Figure 6 bear witness that all the characterized tallows take a higher thermal stability than soybean oil with maximum thermal degradation rates at temperatures higher than those observed for soybean oil. The acetone-extracted tallow showed the lowest thermal stability. The thermal instability of fats, and their derived biodiesel, is related to the degree of unsaturation of aliphatic carbon chains which are converted into hydroperoxides [fifty]. The highest thermal stability of animal fat biodiesel is well documented in the literature [51]. The elapsing of the microwave extractive process did not affect the thermal stability of the extracted fat.
The degree of unsaturation of fatty is relevant for the cetane number of the produced biodiesel but it is as well important for the fat oxidation during the rendering procedure. A higher degree of unsaturation implies lower oxidative stability [52] of fatty and biodiesel [51] because the oxidation process starts with the oxygen attack on the -C=C- bonds [53]. The major products of fatty oxidation contain hydroxyl, perohydroxyl, aldehydes, ketones, carboxylic acids, and trans double bonds, which can be characterized past FTIR. The formation of carboxylic acids during rendering is highly undesired because acids will react with the basic catalysts, leading to soap formation [38]. ATR-FTIR was used to narrate the extracted fats and soybean oil and bands were assigned based on data from the literature. The extracted fats showed intense reflectance bands in the range 1780–1690 cm−1, which are ascribable to the -C=O ester (≈1746 cm−i, [54]) and -C=O carboxylic acrid (1710–1690 cm−1, [55]) vibration modes (Figure 7).
For soybean oil, the ring in this spectral range is elementary and centered at 1744 cm−1 (ester -C=O) with a slight displacement towards smaller wavenumbers, which is uniform with its low acidity (0.66%). The boiling water-extracted tallow shows an analogous band around 1744 cm−1 with a shoulder effectually 1725 cm−1 coming from FFA content (Table two), which is higher than that of soybean oil. The fats extracted with MW show a C=O band broader than soybean oil and boiling water tallow and slightly displaced towards lower wavenumbers, which seems to indicate the formation of ketone and aldehyde species during rendering. Increasing the MW processing time, the spectral shoulder due to carboxylic acids becomes more than intense, thus confirming the oxidizing outcome of microwaves. The acetone-extracted fatty shows a complex FTIR characteristic in the 1780–1690 cm−1 range, with an intense band at 1710 cm−1, which is compatible with its higher acidity (Table 2). Moreover, the acetone-extracted tallow presents a deportation toward smaller wavenumbers in the 2990–2810 cm−i range (-CH2- and -CHthree- vibration modes) (Figure 7), which could exist related to their higher degree of unsaturation and a dissimilar FFA chain length [56]. The FTIR ring around 722 cm−one, belonging to cis-substituted -C=C-, almost vanishes for MW-extracted tallows.
The extracted fats were used to produce biodiesel by methanolysis over the Ca catalyst described above. As previously reported [36], to diminish the undesired fat acidity and moisture furnishings, co-processing with comestible grade soybean oil and solvent-assisted methanolysis (acetone) tests were carried out.
The data in Figure 8 show that the MW- and BW-extracted fats behave similarly in the methanolysis reaction merely lead to a lower FAME yield than that of soybean oil (61.iv–61.7% instead of 88.4%). This activity disuse when tallows were candy is due to their acidity and moisture (Table two). Co-processing BW tallow with SBO (l% Due west/W) allows the partial mitigation of the fat acidity (FAME yield raises from 61.4% to 74.5%). The maximum catalytic activeness using tallow was attained in solvent-assisted (acetone) methanolysis tests for equimixtures of tallow and soybean oil. The acetone-extracted tallow shows similar behavior to the other extracted fats since a mixture containing 25% of acetone-extracted and microwave-extracted fats led to a FAME yield of lxxx%. The methanolysis reaction seems to be indifferent to the unsaturation degree of the processed fat. This result disagrees with that reported past Yang et al. [57]. For homogeneous catalysis with KOH, the authors state that unsaturated fats conduct better during methanolysis because the limiting step is the conversion of diglycerides into monoglycerides, which is faster for polyunsaturated fat molecules. It seems that, in a heterogeneous catalyzed process, the limiting pace of the machinery is unlike to that identified past Yang et al. [57]. Dias et al. [42] emphasize methanol activation every bit the rate-limiting step in the oil methanolysis catalyzed by heterogeneous Ca catalysts.
The data in Figure 8 show that calcium catalysts are promising for biodiesel production from tallow. Data from the literature (Table three) [xix] prove, for dissimilar catalysts, reaction conditions, either similar or more than desperate, to those used to obtain like FAME yields, even using homogeneous catalysts that allow higher reaction rates but which cannot be recovered and reused later the reaction.
All the processed fats allowed the co-product of glycerin with a low level of contamination with species from the reaction medium. The glycerin FTIR spectra in Figure 9 show a low content of MONG (Matter Organic Non-Glycerin [58]) for all the characterized samples, thus indicating a nothing effect of the fatty extraction procedure on the co-produced glycerin purity.
4. Conclusions
Biodiesel was produced from beefiness tallow over a calcium-based heterogeneous catalyst. The raw fatty, collected at a local butcher, was subjected to different rendering processes to split the fat from the tissues. Fat rendering was accomplished using boiling water, microwave-assisted, and acetone extraction at reflux temperature. The boiling water process led to a fatty material with a loftier moisture content (15%) and with the highest energy consumption. The microwave-assisted rendering promoted the slight oxidation of the extracted fat but with high yields (60% instead of 30% for boiling and 15% for acetone processes, by weight) and brusk processing times (3 min instead of forty min in the boiling water procedure). The acetone-extracted fatty showed the highest acidity which can arise from fat hydrolysis during the extraction process. All the extracted fats displayed similar behaviors in the methanolysis reaction (with FAME yields slightly higher than 60%, instead of 88% for pure SBO) over calcium-based catalysts with a decay of catalyst performances due to fatty acidity and moisture. Such undesired effects were decreased by co-processing the extracted beef tallow with soybean oil and using acetone solvent. The data testify that microwave-assisted rendering has a high potential for animal fat processing in biodiesel production. Moreover, since the separated tissues are free of chemicals, they tin can be used in brute food production.
Writer Contributions
Conceptualization, A.P.Due south.D.; methodology, A.P.S.D. and M.R.; validation, A.P.South.D. and B.R.; formal analysis, A.P.S.D. and B.R.; investigation, Yard.R.; resources, A.P.Southward.D.; data curation, B.R.; writing—original draft preparation, A.P.Due south.D., M.R. and B.R.; writing—review and editing, A.P.S.D., Yard.R. and B.R; supervision, A.P.S.D. projection administration, A.P.South.D.; funding acquisition, A.P.Southward.D. All authors have read and agreed to the published version of the manuscript.
Funding
This inquiry was partially funded by FCT (Fundação para a Ciência due east Tecnologia, Portugal) project PTDC/EMS-ENE/4865/2014.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Additional data to those available in the manuscript tin can be provided if requested.
Acknowledgments
The authors admit FCT (Fundação para a Ciência due east Tecnologia, Portugal) for funding project PTDC/European monetary system-ENE/4865/2014.
Conflicts of Involvement
The authors declare that they have no known competing financial interests or personal relationships that could accept appeared to influence the work reported in this paper.
References
- Shan, R.; Zhao, C.; Lv, P.; Yuan, H.; Yao, J. Catalytic applications of calcium rich waste materials for biodiesel: Electric current state and perspectives. Energy Convers. Manag. 2016, 127, 273–283. [Google Scholar] [CrossRef]
- Luque, R.; Lovett, J.C.; Datta, B.; Clancy, J.; Campelo, J.M.; Romero, A.A. Biodiesel equally feasible petrol fuel replacement: A multidisciplinary overview. Energy Environ. Sci. 2010, 3, 1706–1721. [Google Scholar] [CrossRef]
- Gasparatos, A.; Stromberg, P.; Takeuchi, Grand. Sustainability impacts of first-generation biofuels. Anim. Front. 2013, 3, 12–26. [Google Scholar] [CrossRef]
- Chong, C.T.; Loe, T.Y.; Wong, Yard.Y.; Ashokkumar, V.; Lam, South.South.; Chong, Due west.T.; Borrion, A.; Tian, B.; Ng, J.H. Biodiesel sustainability: The global impact of potential biodiesel production on the free energy–water–food (EWF) nexus. Environ. Technol. Innov. 2021, 22, 101408. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, X.; Adamu, A.; Zhu, Z. Economic evaluation and product process simulation of biodiesel production from waste cooking oil. Curr. Res. Green Sustain. Chem. 2021, iv, 100091. [Google Scholar] [CrossRef]
- Festel, G.; Bellof, M.; Würmseher, Chiliad.; Rammer, C.; Boles, E. Calculation of raw material prices and conversion costs for biofuels. Lect. Notes Energy 2014, 27, 93–115. [Google Scholar] [CrossRef]
- Bashir, Thousand.A.; Wu, S.; Zhu, J.; Krosuri, A.; Khan, M.U.; Ndeddy Aka, R.J. Recent evolution of advanced processing technologies for biodiesel product: A critical review. Fuel Process. Technol. 2022, 227, 107120. [Google Scholar] [CrossRef]
- Sander, A.; Antonije Košćak, Yard.; Kosir, D.; Milosavljević, N.; Parlov Vuković, J.; Magić, L. The influence of animal fat type and purification conditions on biodiesel quality. Renew. Energy 2018, 118, 752–760. [Google Scholar] [CrossRef]
- Habib, M.S.; Tayyab, One thousand.; Zahoor, S.; Sarkar, B. Management of animal fat-based biodiesel supply chain under the prototype of sustainability. Energy Convers. Manag. 2020, 225, 113345. [Google Scholar] [CrossRef]
- UFOP Report on Global Marketplace Supply European and World Demand for Biomass for the Purpose of Biofuel Production in Relation to Supply in the Nutrient and Feedstuff Markets; UFOP: Ouro Preto, Brazil, 2018.
- Stojković, I.J.; Miladinović, M.R.; Stamenković, O.South.; Banković-Ilić, I.B.; Povrenović, D.S.; Veljković, V.B. Biodiesel production by methanolysis of waste lard from piglet roasting over quicklime. Fuel 2016, 182, 454–466. [Google Scholar] [CrossRef]
- Mata, T.M.; Mendes, A.Chiliad.; Caetano, Northward.Southward.; Martins, A.A. Properties and sustainability of biodiesel from animal fats and fish oil. Chem. Eng. Trans. 2014, 38, 175–180. [Google Scholar]
- Vafakish, B.; Barari, M. Biodiesel product by transesterification of tallow fat using heterogeneous Catalysis. Kem. Ind. 2017, 66, 47–52. [Google Scholar] [CrossRef]
- Banković-Ilić, I.B.; Stojković, I.J.; Stamenković, O.Due south.; Veljkovic, V.B.; Hung, Y.T. Waste material animate being fats as feedstocks for biodiesel production. Renew. Sustain. Energy Rev. 2014, 32, 238–254. [Google Scholar] [CrossRef]
- Ramos, One thousand.; Dias, A.P.South.; Puna, J.F.; Gomes, J.; Bordado, J.C. Biodiesel production processes and sustainable raw materials. Energies 2019, 12, 4408. [Google Scholar] [CrossRef]
- Adewale, P.; Dumont, M.J.; Ngadi, M. Recent trends of biodiesel product from fauna fat wastes and associated production techniques. Renew. Sustain. Energy Rev. 2015, 45, 574–588. [Google Scholar] [CrossRef]
- Wyatt, V.T.; Hess, One thousand.A.; Dunn, R.O.; Foglia, T.A.; Haas, G.J.; Marmer, W.N. Fuel properties and nitrogen oxide emission levels of biodiesel produced from animal fats. J. Am. Oil Chem. Soc. 2005, 82, 585–591. [Google Scholar] [CrossRef]
- Ikura, M.; Kouchachvili, 50.; Caravaggio, One thousand. Production of biodiesel from waste fat and grease. Lect. Notes Energy Environ. 2007, 25–30. [Google Scholar]
- Toldrá-Reig, F.; Mora, L.; Toldrá, F. Trends in biodiesel product from animal fatwaste. Appl. Sci. 2020, 10, 3644. [Google Scholar] [CrossRef]
- Woodgate, S.; van der Veen, J. The role of fat processing and rendering in the European Matrimony animal production industry. Biotechnol. Agron. Soc. Environ. 2004, 8, 283–294. [Google Scholar]
- Meeker, D.50. ESSENTIAL Rendering All Almost The Fauna By-Products Industry; The National Renderers Association The Fats and Proteins Research Foundation The Creature Protein Producers Industry: Alexandria, VA, USA, 2006; ISBN 0965466035. [Google Scholar]
- Jayathilakan, K.; Sultana, K.; Radhakrishna, Thou.; Bawa, A.Due south. Utilization of byproducts and waste material materials from meat, poultry and fish processing industries: A review. J. Nutrient Sci. Technol. 2012, 49, 278–293. [Google Scholar] [CrossRef] [PubMed]
- Kazimierowicz, J.; Zieliński, One thousand.; Dębowski, M. Influence of the heating method on the efficiency of biomethane production from expired food products. Fermentation 2021, 7, 12. [Google Scholar] [CrossRef]
- Zieliński, 1000.; Dębowski, K.; Kazimierowicz, J. The effect of electromagnetic microwave radiation on marsh gas fermentation of selected energy ingather species. Procedure 2022, 10, 45. [Google Scholar] [CrossRef]
- Jákói, Z.; Lemmer, B.; Hodúr, C.; Beszédes, S. Microwave and ultrasound based methods in sludge treatment: A review. Appl. Sci. 2021, 11, 7067. [Google Scholar] [CrossRef]
- Quesada-Salas, M.C.; Delfau--bonnet, G.; Willig, Thou.; Préat, N.; Allais, F.; Ioannou, I. Optimization and comparison of three prison cell disruption processes on lipid extraction from microalgae. Procedure 2021, nine, 369. [Google Scholar] [CrossRef]
- Aguado-Deblas, 50.; Estevez, R.; Russo, M.; La Parola, Five.; Bautista, F.M.; Testa, M.L. Microwave-assisted glycerol etherification over sulfonic acrid catalysts. Materials 2020, 13, 1584. [Google Scholar] [CrossRef] [PubMed]
- Nomanbhay, Due south.; Ong, M.Y. A review of microwave—Assisted reactions for biodiesel production. Bioengineering 2017, 4, 57. [Google Scholar] [CrossRef]
- Sharma, H.; Giriprasad, R.; Goswami, M. Fauna fat-processing and its quality control. J. Food Process. Technol. 2013, 4, 8. [Google Scholar] [CrossRef]
- Lin, L.; Liu, D.; Chen, M.; Kuo, H.; Tan, F. Influence of rendering methods on the yield and quality characteristics of craven fat from broiler skin fat. Asian-Australas J. Anim. Sci. 2013, xxx, 2011–2013. [Google Scholar]
- Sheu, K.S.; Chen, T.C. Yield and quality characteristics of edible broiler pare fat as obtained from 5 rendering methods. J. Nutrient Eng. 2002, 55, 263–269. [Google Scholar] [CrossRef]
- Hewavitharana, G.1000.; Perera, D.N.; Navaratne, S.B.; Wickramasinghe, I. Extraction methods of fatty from food samples and preparation of fatty acid methyl esters for gas chromatography: A review. Arab. J. Chem. 2020, 13, 6865–6875. [Google Scholar] [CrossRef]
- Balasubramanian, Due south.; Allen, J.D.; Kanitkar, A.; Boldor, D. Oil extraction from Scenedesmus obliquus using a continuous microwave system—design, optimization, and quality label. Bioresour. Technol. 2011, 102, 3396–3403. [Google Scholar] [CrossRef]
- Atabani, A.Eastward.; Silitonga, A.S.; Badruddin, I.A.; Mahlia, T.Thou.I.; Masjuki, H.H.; Mekhilef, S. A comprehensive review on biodiesel as an culling energy resource and its characteristics. Renew. Sustain. Energy Rev. 2012, sixteen, 2070–2093. [Google Scholar] [CrossRef]
- Mahmudul, H.Yard.; Hagos, F.Y.; Mamat, R.; Adam, A.A.; Ishak, Due west.F.Westward.; Alenezi, R. Production, label and operation of biodiesel equally an alternative fuel in diesel fuel engines—A review. Renew. Sustain. Energy Rev. 2017, 72, 497–509. [Google Scholar] [CrossRef]
- Dias, A.P.S.; Ramos, M.; Catarino, Grand.; Puna, J.; Gomes, J. Towards biodiesel sustainability: Solvent assisted biodiesel product by co-processing beefiness tallow and soybean oil over calcium catalysts. Energy Fuels 2020, xi, 6249–6259. [Google Scholar]
- Marwaha, A.; Rosha, P.; Mohapatra, South.K.; Mahla, South.Grand.; Dhir, A. Waste matter materials as potential catalysts for biodiesel production: Electric current state and hereafter scope. Fuel Process. Technol. 2018, 181, 175–186. [Google Scholar] [CrossRef]
- Soares Dias, A.P.; Puna, J.; Neiva Correia, M.J.; Nogueira, I.; Gomes, J.; Bordado, J. Consequence of the oil acerbity on the methanolysis performances of lime goad biodiesel from waste material frying oils (WFO). Fuel Process. Technol. 2013, 116, 94–100. [Google Scholar] [CrossRef]
- EN 14214:2003; Automotive Fuels—Fat acid Methyl Esters (FAME) for Diesel Engines—Requirements. European Commission for Standardization (Comité Européen de Normalisation): Brussel, Belgium, 2003.
- Dias, A.P.S.; Puna, J.; Gomes, J.; Ramos, M.; Rijo, B.; Bordado, J. The role of alkali dopants on the oil methanolysis behavior of lime catalyst: Activity & stability. Energy Sources Part A Recovery Util. Environ. Eff. 2022, 44, 748–757. [Google Scholar] [CrossRef]
- Soares Dias, A.P.; Catarino, One thousand.; Gomes, J. Co-processing lard/soybean oil over Ca-based catalysts to greener biodiesel. Environ. Technol. Innov. 2021, 21, 101220. [Google Scholar] [CrossRef]
- Soares Dias, A.P.; Puna, J.; Gomes, J.; Neiva Correia, M.J.; Bordado, J. Biodiesel production over lime. Catalytic contributions of bulk phases and surface Ca species formed during reaction. Renew. Free energy 2016, 99, 622–630. [Google Scholar] [CrossRef]
- Soares Dias, A.P.; Ramos, M.; Catarino, One thousand.; Costa Pereira, Thou.F. Biodiesel past Co-processing animate being fat/vegetable oil mixtures over basic heterogeneous Ca goad. Clean. Eng. Technol. 2020, 1, 100012. [Google Scholar] [CrossRef]
- Jayakumar, G.; Karmegam, N.; Gundupalli, M.P.; Bizuneh Gebeyehu, Thousand.; Tessema Asfaw, B.; Chang, S.W.; Ravindran, B.; Kumar Awasthi, Thousand. Heterogeneous base of operations catalysts: Synthesis and application for biodiesel production—A review. Bioresour. Technol. 2021, 331, 125054. [Google Scholar] [CrossRef] [PubMed]
- Catarino, M.; Ramos, Thou.; Dias, A.P.S.; Santos, M.T.; Puna, J.F.; Gomes, J.F. Calcium rich food wastes based catalysts for biodiesel production. Waste Biomass Valorization 2017, 8, 1699–1707. [Google Scholar] [CrossRef]
- Dias, A.P.S.; Ramos, G. On the storage stability of CaO biodiesel catalyst. Hydration and carbonation poisoning. J. Environ. Chem. Eng. 2021, 9, 104917. [Google Scholar] [CrossRef]
- Xing, R.; Qin, Y.; Guan, X.; Liu, Southward.; Yu, H.; Li, P. Comparison of antifungal activities of scallop shell, oyster shell and their pyrolyzed products. Egypt. J. Aquat. Res. 2013, 39, 83–xc. [Google Scholar] [CrossRef]
- Odetoye, T.E.; Agu, J.O.; Ajala, Due east.O. Biodiesel production from poultry wastes: Waste product chicken fat and eggshell. J. Environ. Chem. Eng. 2021, 9, 105654. [Google Scholar] [CrossRef]
- Iasmin, M.; Dean, Fifty.O.; Ducoste, J.J. Quantifying fat, oil, and grease deposit germination kinetics. Water Res. 2016, 88, 786–795. [Google Scholar] [CrossRef]
- Demirbas, A. Thermal deposition of fatty acids in biodiesel production by supercritical methanol. Energy Explor. Exploit. 2007, 25, 63–70. [Google Scholar] [CrossRef]
- Saluja, R.K.; Kumar, V.; Sham, R. Stability of biodiesel—A review. Renew. Sustain. Energy Rev. 2016, 62, 866–881. [Google Scholar] [CrossRef]
- Joseph, P. Oxidative Stability and Shelf Life of Majority Animal Fats and Poultry Fats; Elsevier Inc.: Amsterdam, The netherlands, 2016; ISBN 9781630670566. [Google Scholar]
- Borchman, D.; Sinha, S. Determination of products of lipid oxidation by infrared spectroscopy. Methods Mol. Biol. 2002, 186, 21–28. [Google Scholar] [CrossRef]
- Christy, A.A.; Egeberg, P.K. Quantitative decision of saturated and unsaturated fatty acids in edible oils by infrared spectroscopy and chemometrics. Chemom. Intell. Lab. Syst. 2006, 82, 130–136. [Google Scholar] [CrossRef]
- Nina Naquiah, A.North.; Marikkar, J.Thousand.N.; Mirghani, Grand.E.S.; Nurrulhidayah, A.F.; Yanty, N.A.M. Differentiation of fractionated components of lard from other creature fats using different analytical techniques. Sains Malays. 2017, 46, 209–216. [Google Scholar]
- Bobroff, 5.; Rubio, C.; Vigier, V.; Petibois, C. FTIR spectroscopy characterization of fatty-acyl-chain conjugates. Anal. Bioanal. Chem. 2016, 408, 319–326. [Google Scholar] [CrossRef]
- Yang, J.; Astatkie, T.; He, Q.S. A comparative study on the effect of unsaturation caste of camelina and canola oils on the optimization of bio-diesel production. Free energy Rep. 2016, two, 211–217. [Google Scholar] [CrossRef]
- Catarino, K.; Ferreira, E.; Soares Dias, A.P.; Gomes, J. Dry washing biodiesel purification using fumed silica sorbent. Chem. Eng. J. 2020, 386, 123930. [Google Scholar] [CrossRef]
Effigy 1. Biodiesel production from animal fat flowsheet (adapted from [xix]).
Effigy 1. Biodiesel production from animal fatty flowsheet (adapted from [19]).
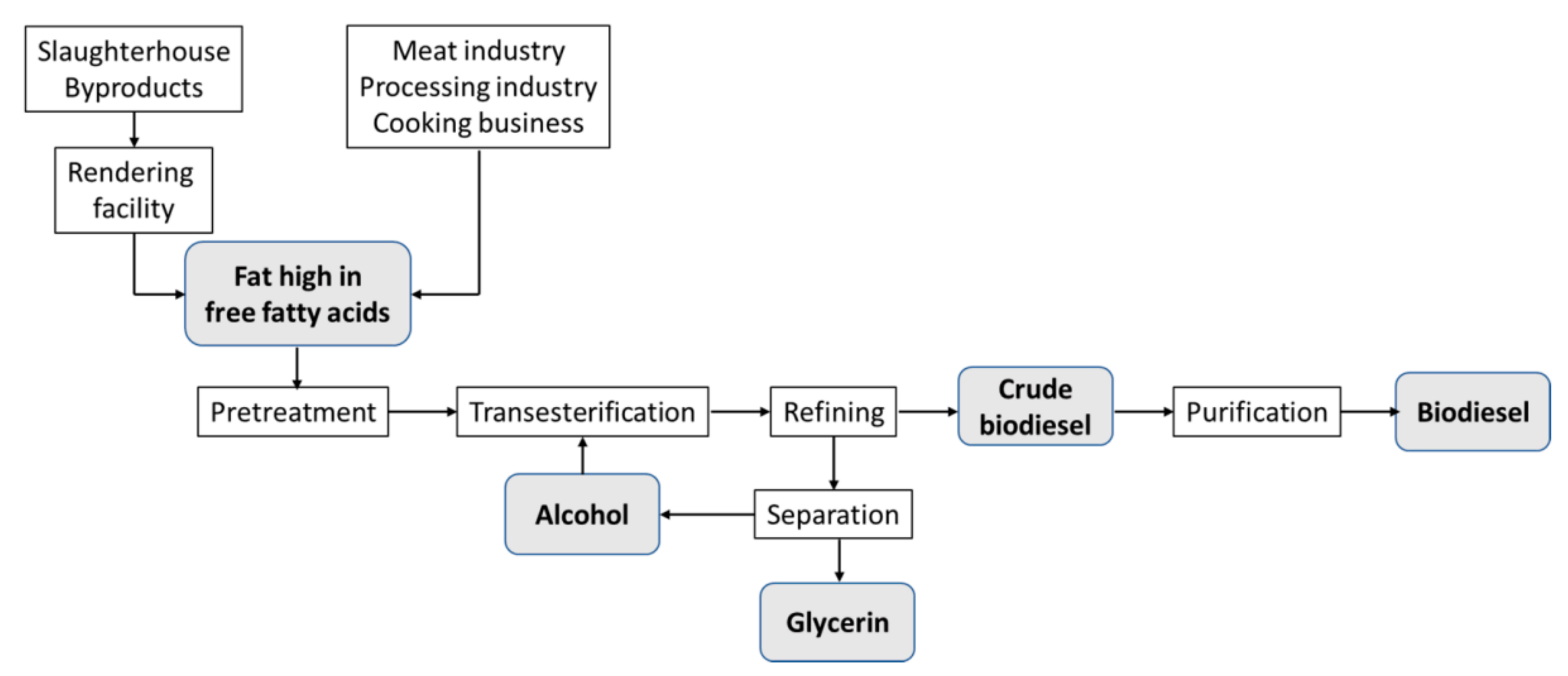
Figure 2. Experimental part flowchart.
Figure 2. Experimental part flowchart.
Figure 3. SEM micrographs of raw and calcined scallop shells.
Figure 3. SEM micrographs of raw and calcined scallop shells.

Figure 4. XRD pattern of fresh catalysts (scallop shell calcined at 800 °C) and JCPDS standard files for calcite (CaCOthree) and lime (CaO).
Figure 4. XRD pattern of fresh catalysts (scallop shell calcined at 800 °C) and JCPDS standard files for calcite (CaCOiii) and lime (CaO).

Effigy five. FTIR spectrum of the as-prepared catalyst (scallop shells calcined for 3 h at 800 °C).
Figure 5. FTIR spectrum of the as-prepared catalyst (scallop shells calcined for 3 h at 800 °C).

Figure half-dozen. Thermal stability and moisture content of the extracted tallows and soybean oil, assessed by thermogravimetry.
Figure vi. Thermal stability and moisture content of the extracted tallows and soybean oil, assessed past thermogravimetry.
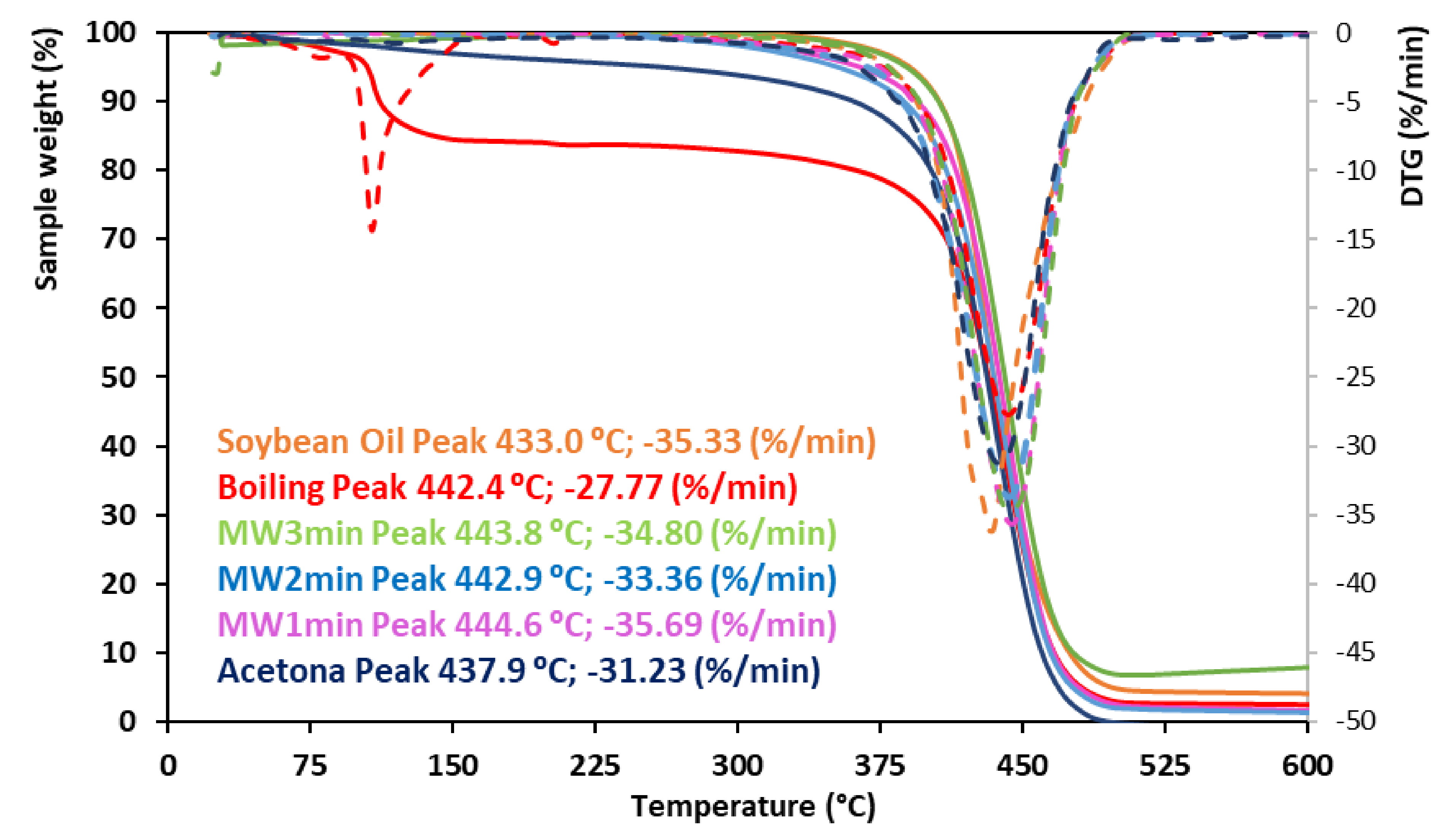
Figure 7. ATR-FTIR spectra of tallows, extracted using the various procedures, with SBO every bit a standard.
Figure 7. ATR-FTIR spectra of tallows, extracted using the diverse procedures, with SBO every bit a standard.
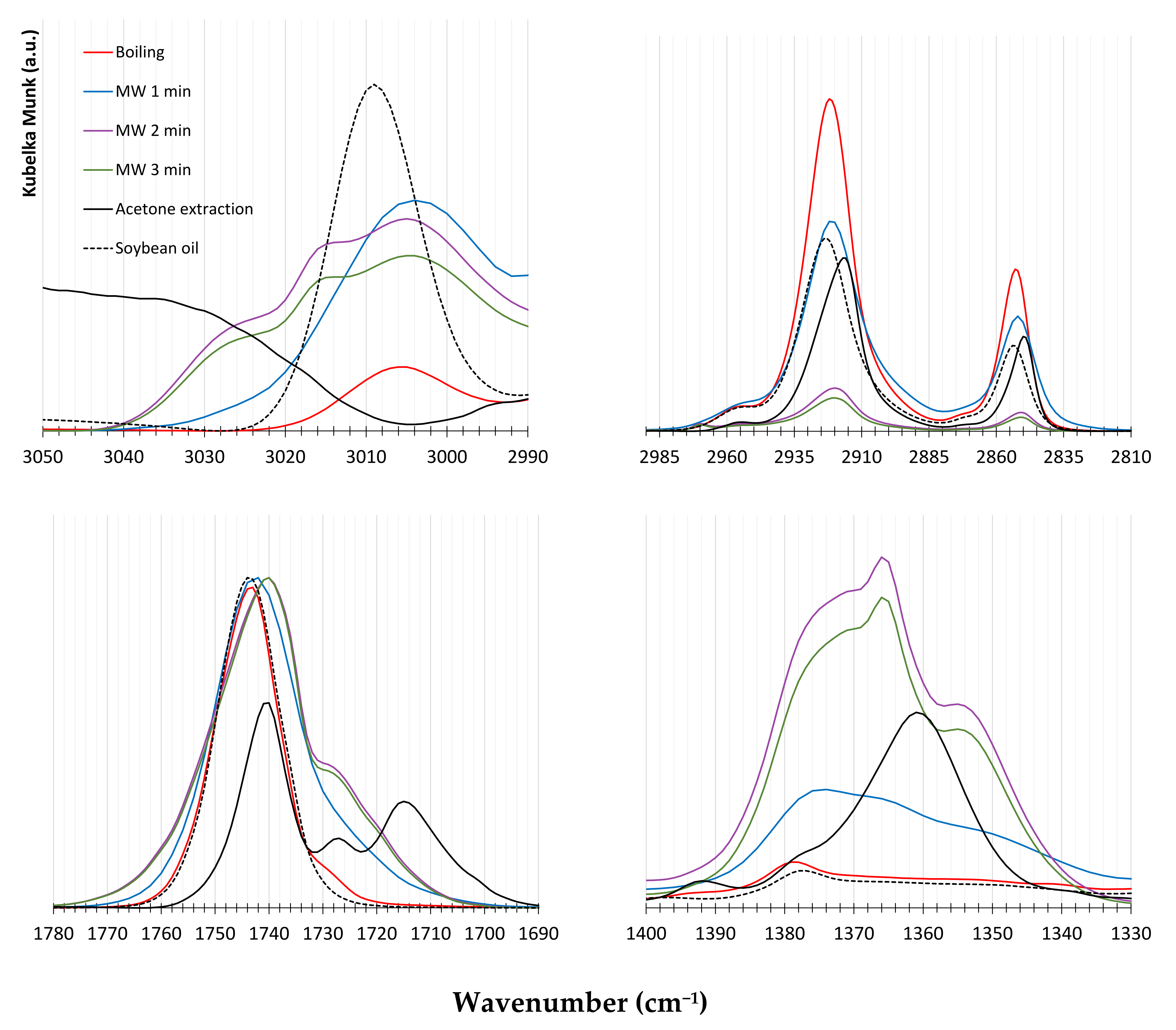
Figure 8. FAME yield obtained past methanolysis of the extracted fats at reflux temperature (methanol/oil = 12 molar ratio; Wcat/Woil = 5%; two.5 h reaction time). The methanolysis of soybean oil (SBO) was used as a standard.
Effigy eight. FAME yield obtained by methanolysis of the extracted fats at reflux temperature (methanol/oil = 12 molar ratio; Wcat/Due westoil = v%; 2.v h reaction time). The methanolysis of soybean oil (SBO) was used as a standard.

Effigy 9. ATR-FTIR spectra of glycerin coproduced with biodiesel and commercial glycerin used every bit standard.
Effigy 9. ATR-FTIR spectra of glycerin coproduced with biodiesel and commercial glycerin used as standard.
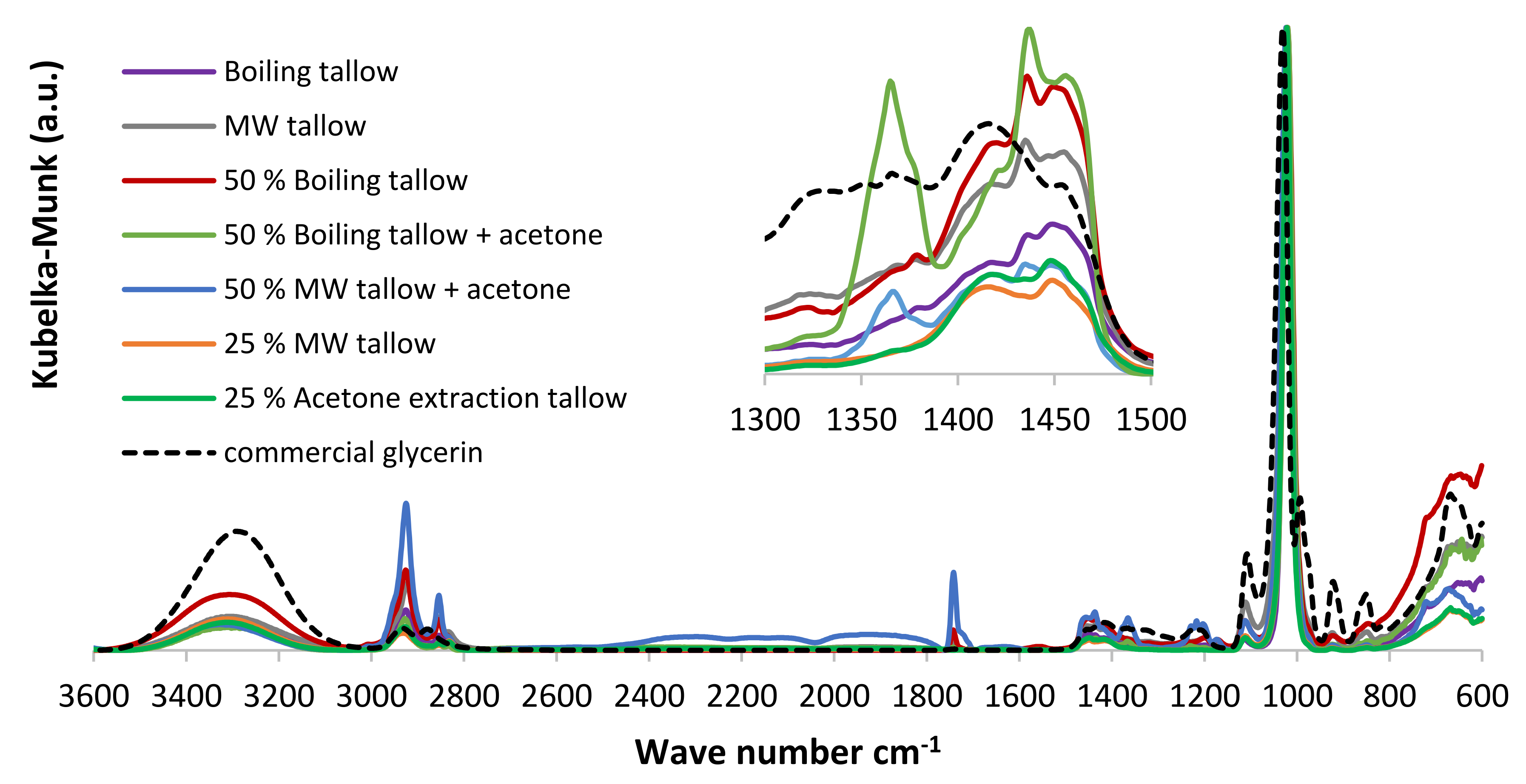
Table 1. Fats raw materials prices (world wide web.alibaba.com, accessed on i March 2022).
Tabular array 1. Fats raw materials prices (www.alibaba.com, accessed on 1 March 2022).
| Fatty | EUR per Metric Ton |
|---|---|
| Soybean oil | 184–450 |
| Rapeseed oil | 230–583 |
| Palm oil (rough) | 138–238 |
| UCO | 137–200 |
| Tallow (category one) | 100–148 |
Table two. Energy consumed in rendering processes, fatty yield, and fat acidity.
Tabular array 2. Free energy consumed in rendering processes, fat yield, and fat acidity.
| Rendering Procedure | Equipment | Ability (W) | Time (min) | Energy (kWh/100 yard Raw Fat) | Fat Yield (g/100 g Raw Fatty) | Fat Acidity (%) |
|---|---|---|---|---|---|---|
| Microwave | MW oven | 1400 | three | 0.070 | 60 ± iii | 0.81 |
| Boiling water | Stove | 2800 | forty | 1.868 | 30 * ± 3 | 1.96 |
| Acetone extraction | Heating mantle | 325 | 240 | 1.300 | 15 ± 1 | ii.39 |
Tabular array three. FAME yields from tallow using dissimilar catalysts (data collected from [19]).
Table three. FAME yields from tallow using different catalysts (data nerveless from [nineteen]).
| Catalyst | %Wcat (Fat Basis) | Methanol/Fat (Molar Ratio) | Reaction Temperature; Time | FAME Yield (%) |
|---|---|---|---|---|
| HtwoSOfour | 1.25 | 30 | 60 °C; 24 h | 93.2 |
| KOH | 0.viii | vi | sixty °C; 2 h | xc.8 |
| Immobilized lipase | 20 | 12 | 50 °C; 48 h | 89.7 |
| Publisher'south Notation: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open admission article distributed under the terms and conditions of the Creative Eatables Attribution (CC By) license (https://creativecommons.org/licenses/by/4.0/).
Source: https://www.mdpi.com/2227-9717/10/4/666/htm
0 Response to "Beef Fat Extract Beef Fat Extract Ole"
Post a Comment